Product Description
The Filariasis IgG/IgM Test Kit (Colloidal Gold) is a lateral flow immuno assay for the simultaneous detection and differentiation of IgG and IgM anti-lymphatic filarial parasites (W. Bancrofti and B. Malayi) in human serum, plasma or whole blood. This test is intended to be used as a screening test and as an aid in the diagnosis of infection with lymphatic filarial parasites.
【Principles of the Test 】
The Filariasis IgG/IgM Test Kit (Colloidal Gold) is a lateral flow chromatographic immunoassay.
The test card contains:
1. Colloidal gold-labeled antigen and quality control antibody complex.
2. Nitrocellulose membranes immobilized with two test lines (M line and G line) and one quality control line (C line).
When an appropriate amount of sample is added to the sample well of the test card, the sample will move forward along the test card under capillary action.
W. bancrofti or B. malayi IgM antibodies if present in the specimen willbind to the Filariasis conjugates. The immunocomplex is then captured on the membrane by the pre-coated anti-human IgM antibody, forming a burgundy colored M line, indicating a W. bancrofti or B. malayi IgM positive test result.
W.bancrofti or B. malayi IgG antibodies if present in the specimen will bind to the Filariasis conjugates. The immunocomplex is then captured by the pre-coated reagents on the membrane, forming a burgundy colored G line, indicating a W. bancrofti or B. malayi IgG positive test result.
Absence of any test lines (M and G) suggests a negative result. The test contains an internal control (C line) which should exhibit a burgundy colored line of the immunocomplex conjugate regardless of the color development on any of the test lines.Otherwise, the test result is invalid and the specimen must be retested with another device.
【Reagents And Materials Supplied】
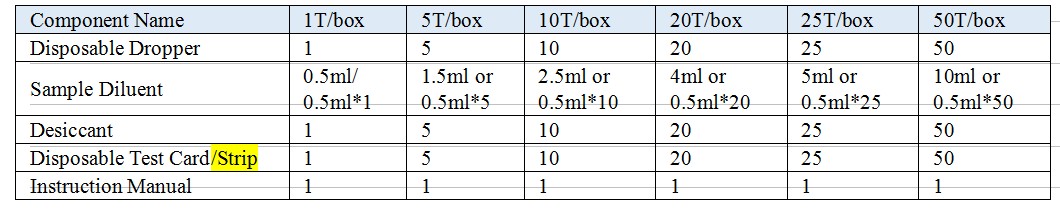
Model: Test Card, Test Strip
【Test Procedure】
1.Before opening the bag, please leave it at room temperature. Take the test device out of the sealed bag and use it as soon as possible. The best results will be obtained if the measurement is performed within one hour.
2.Dispense 35 µL of serum/plasma or whole blood into the sample wells of the test card.
3.Dispense 1 drop of buffer directly from the buffer bottle, or use a calibrated pipette to transfer 40 µL of buffer to the sample well.
4.The result should be between 10 and 20 minutes, but not more than 30 minutes
【Shelf Life and Storage 】
1. Store in a dry place at 2~30°C away from light.
2.Transport at2-37℃ for 20days
3. After opening the inner packaging, the test card will become invalid due to moisture absorption, please use it within 1 hour.
4. The shelf life of the test kit is 12 months from date of manufacture.
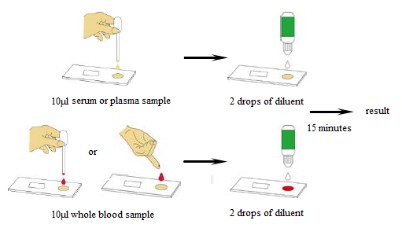
【Test Procedure】
Step 1: Bring the specimen and test components to room temperature if refrigerated or frozen. Mix the specimen well prior to assay once thawed.
Step 2: When ready to test, open the pouch at the notch and remove device. Place the test device on a clean, flat surface.
Step 3: Be sure to label the device with specimen's ID number.
For whole blood test
Apply 1 drop of whole blood (about 40-50 µL) into the sample well. Then add 2 drops (about 70-100 µL) of Sample Diluent immediately.
For serum or plasma test
Fill the pipette dropper with the specimen.
Step 4: Holding the dropper vertically, dispense 1 drop (about 30-45 µL) of specimen into the sample well making sure that there are no air bubbles.
Then add 2 drops (about 70-100 µL) of Sample Diluent immediately.
Step 5: Set up timer.
Step 6: Results can be read in 15 minutes. Positive results can be visible in as short as 1 minute.
Don't read result after 15 minutes. To avoid confusion, discard the test device after interpreting the result.
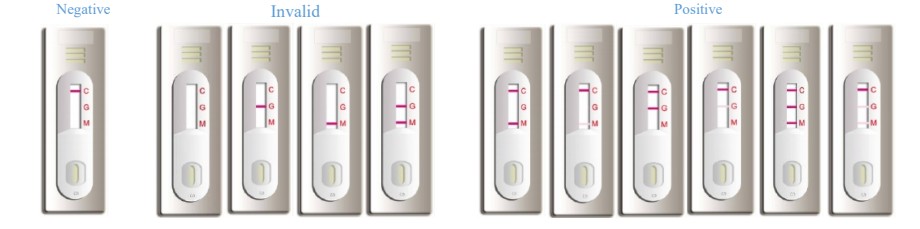
【Interpretation of Results】
practice including adding positive and negative controls in order to verify proper test performance.
NEGATIVE:
If only the quality control line C appears, and the test lines M and G are not purple/red, it indicates that no antibody is detected, and the result is negative.
POSITIVE:
IgM positive: If both the quality control line C and the test line M appear purple/red, it indicates that the Ig M antibody is detected, and the result is positive for Ig M antibody. IgG positive: If both the quality control line C and the test line G appear purple/red, it indicates that the Ig G antibody is detected, and the result is positive for Ig G antibody. IgM and IgG positive: If the quality control line C and the test lines M and G all appear purple/red, it indicates that the Ig M and Ig G antibodies are detected, and the result is positive for both IgM and IgG antibodies.
INVALID:
If the quality control line C is not displayed, the test result is invalid regardless of whether there is a purple/red test line, and it should be tested again.
Online Message
Products Recommended
Please give us a message