Product Description
Babio®Typhoid IgG/IgM Test Kit (Colloidal Gold Method) is a rapid chromatographic immunoassay for the qualitative detection and differentiation of specific IgM and IgG antibodies against specific Salmonella typhi antigen in human serum or plasma. It is intended for in vitro diagnosis of typhoid fever.
【Test Principle】
The Babio® Typhoid IgG/IgM Test Kit (Colloidal Gold Method) is a method for qualitative detection of IgG and IgM antibodies against Streptococcus typhi in human serum, plasma or whole blood. The test provides a differential detection of anti-S. typhi-IgG and anti-S. typhi-IgM antibodies and can be used to presumptive distinction between a current, latent and/or carrier S. typhi infection. Serum, plasma or whole blood samples could have been used for this test. The specific S. typhi antigen is immobilized onto cellulose nitrate membrane as test lines. When the test sample is added to the sample pad, it migrates upwards. If IgG or IgM antibodies to S. typhi are present in the specimen they will bind to the colloidal gold-antigen conjugate. The complex will continue to move on the cellulose nitrate membrane and then captured at the test window zone by the immobilized specific S. typhi antigen, and form pale to dark lines. The intensity of the lines will vary depending on the amount of antibody present in the sample. The appearance of a colored line in a specific test region should be considered as positive for that particular antibody (IgG and/or IgM). As a control, a colored line will always appear in the control line area, indicating that the appropriate specimen volume and the appropriate membrane wick have been added.
【Reagents And Materials Supplied】
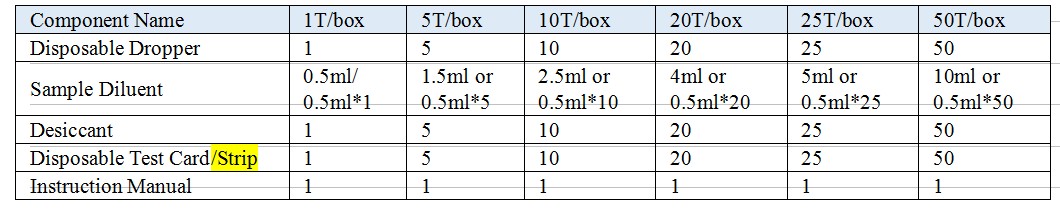
Model: Test Card, Test Strip
【Test Procedure】
1.Before opening the bag, please leave it at room temperature. Take the test device out of the sealed bag and use it as soon as possible. The best results will be obtained if the measurement is performed within one hour.
2.Dispense 35 µL of serum/plasma or whole blood into the sample wells of the test card.
3.Dispense 1 drop of buffer directly from the buffer bottle, or use a calibrated pipette to transfer 40 µL of buffer to the sample well.
4.The result should be between 10 and 20 minutes, but not more than 30 minutes
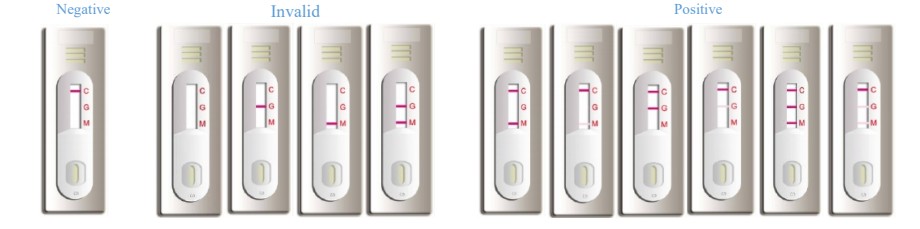
【Interpretation of Results】
practice including adding positive and negative controls in order to verify proper test performance.
NEGATIVE:
If only the quality control line C appears, and the test lines M and G are not purple/red, it indicates that no antibody is detected, and the result is negative.
POSITIVE:
IgM positive: If both the quality control line C and the test line M appear purple/red, it indicates that the Ig M antibody is detected, and the result is positive for Ig M antibody. IgG positive: If both the quality control line C and the test line G appear purple/red, it indicates that the Ig G antibody is detected, and the result is positive for Ig G antibody. IgM and IgG positive: If the quality control line C and the test lines M and G all appear purple/red, it indicates that the Ig M and Ig G antibodies are detected, and the result is positive for both IgM and IgG antibodies.
INVALID:
If the quality control line C is not displayed, the test result is invalid regardless of whether there is a purple/red test line, and it should be tested again.
【Limitations】
Read the package insert in its entirety prior to performing the test. Failure to follow the package insert instructions may result in an invalid test result.
2. This product is for qualitative assessment only. This test is only provided for use by clinical laboratories or to health care workers for point of care testing.
3. As with all diagnostic tests, all results must be interpreted together with other clinical information available to the physician.
4. Negative results do not preclude Typhoid infection and should not be used as the sole basis for patient management decisions. Follow-up testing with a molecular diagnostic should be considered to rule out infection in these individuals.
5. Due to the limitation of detection sensitivity, negative results may be caused by antibody concentrations lower than the analytical sensitivity of the product.
6. This test will only indicate the presence of Typhoid IgM and/or IgG antibodies in the specimen.
7. Some specimens containing unusually high titer of heterophile antibodies or rheumatoid factor may affect expected results.
8. Not for screening of donated blood.
Online Message
Products Recommended
Please give us a message